 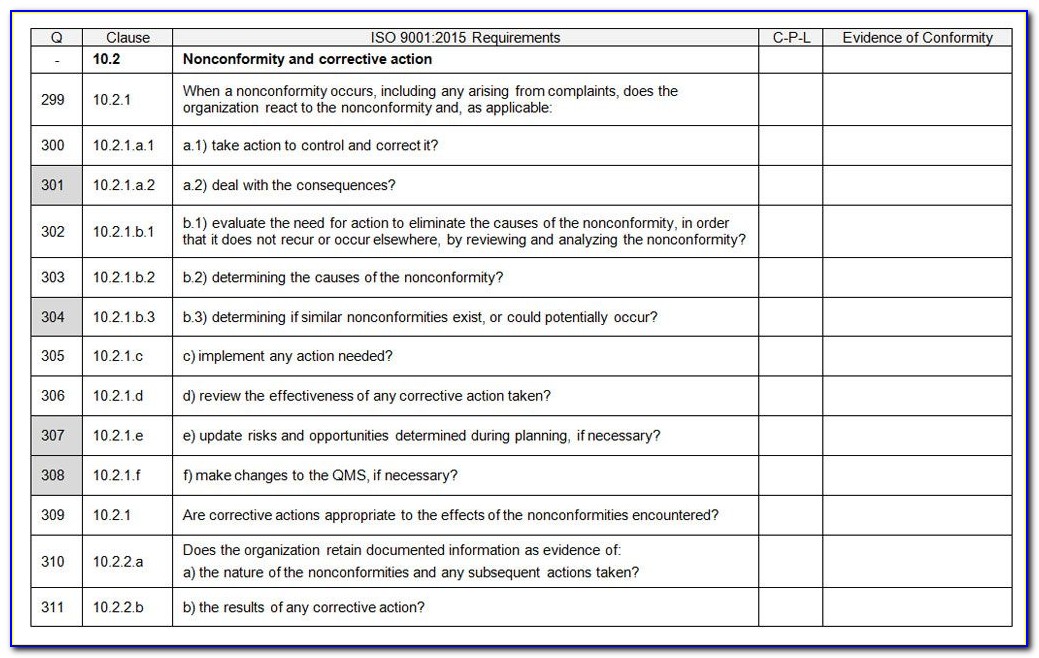
Internal Audit: This is used to test initial compliance.This necessitates comprehensive policies, processes, work instructions, records, and other documents. 
Documentation: The applicant team must document how their QMS meets the standard.This need support from the entire organization. This often needs a significant cultural transformation within the firm. Preparation: The applicant must create and implement a QMS that meets the standards.The ISO 13485 auditing and accreditation process typically includes the following steps: ISO 13485’s Audit and Certification Process Management Review: Auditors examine top management’s internal QMS evaluation and improvement strategies.The organization’s process for addressing these issues may also be evaluated. 
The non-compliant components will then have to be re-audited.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
